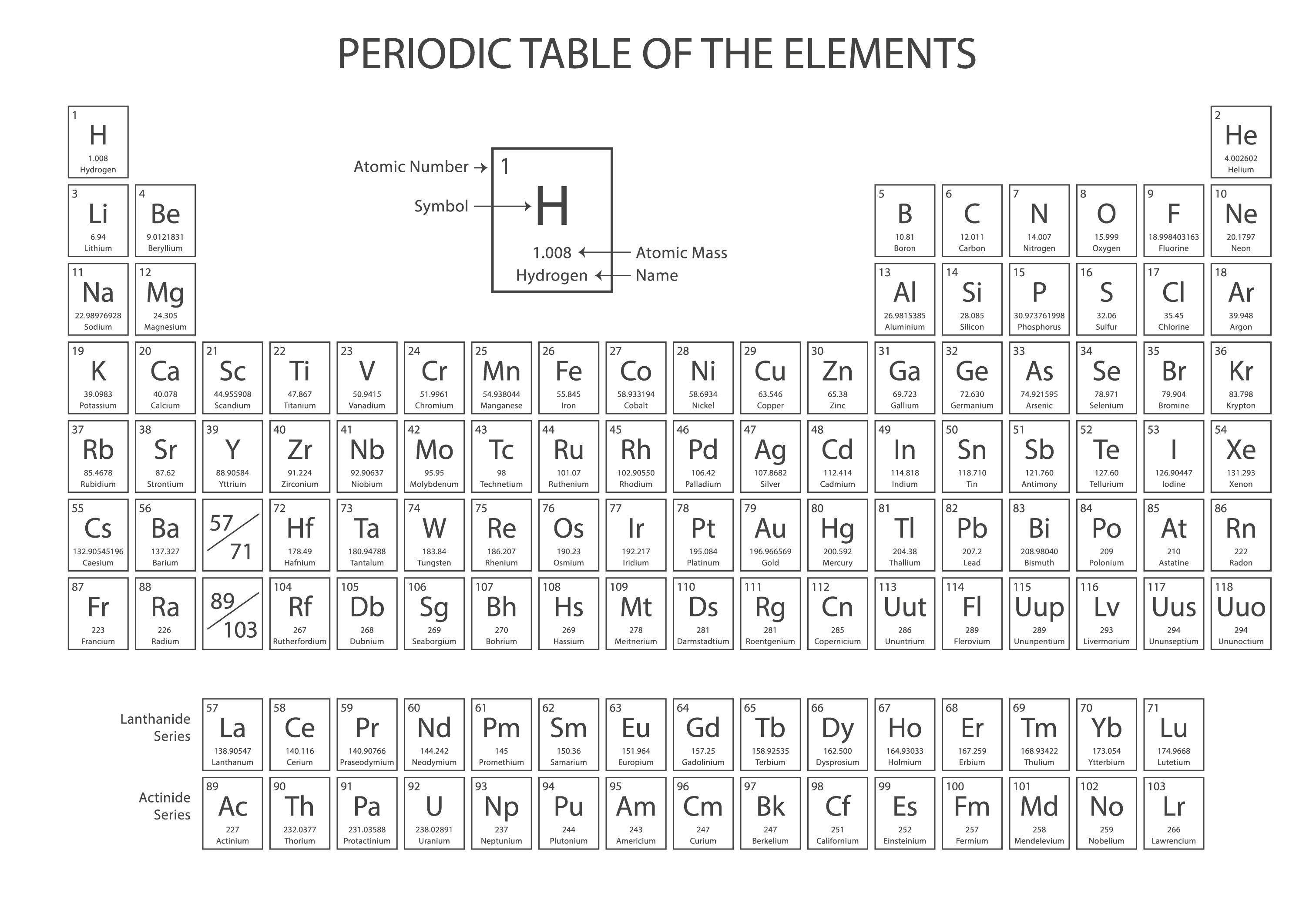
What is the atomic number of an element whose atoms each contain 47 protons, 60 neutrons, and 47 electrons? (1) 13 (3) 60 (2) 47 (4) 107 _12. What is the atomic number of an element that has six protons and eight neutrons? (1) 6 (3) 8 (2) 2 (4) 14 _11. What is the charge of the nucleus in an atom of oxygen-17? (1) 0 (3) +8 (2) –2 (4) +17 _10. What is the nuclear charge of an atom with a mass of 23 and an atomic number of 11? (1) 11+ (3) 23+ (2) 12+ (4) 34+ _8.
#The periodic table chemistry test wizar plus#
All atoms of a given element must contain the same number of (1) protons (2) neutrons (3) electrons plus neutrons (4) protons plus neutrons _7. Which subatomic particle has a negative charge? (1) proton (3) neutron (2) electron (4) positron Quarterly 1 Review _6. Which of the following particles has the smallest mass? (1) neutron (3) proton (2) electron (4) hydrogen atom _5.
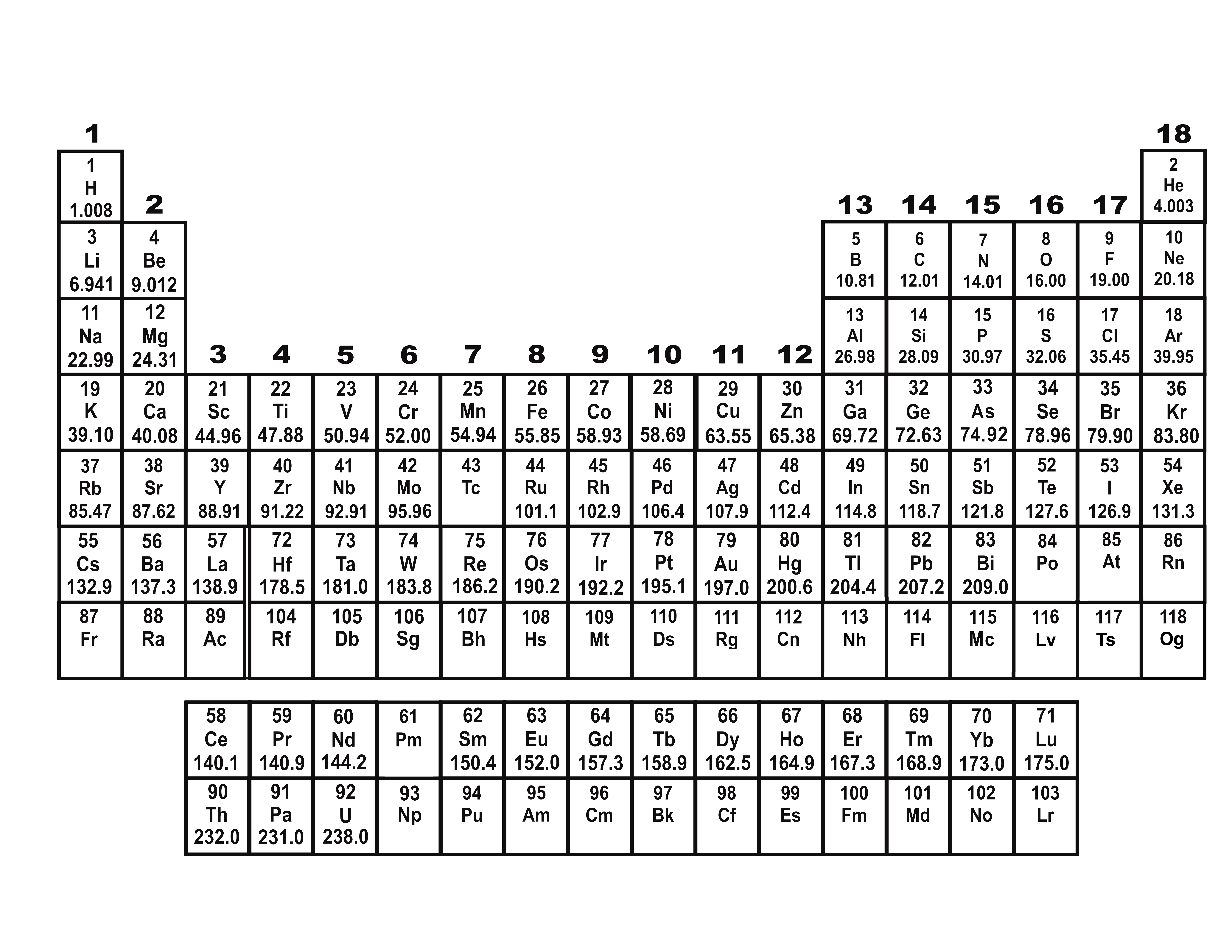
Experiments performed to reveal the structure of atoms led scientists to conclude that an atom's (1) positive charge is evenly distributed throughout its volume (2) negative charge is mainly concentrated in its nucleus (3) mass is evenly distributed throughout its volume (4) volume is mainly unoccupied _4. (4) Alpha particles are more dense than gold atoms. (3) Alpha particles and gold nuclei have opposite charges. (2) Gold atoms are similar to alpha particles. Which statement about gold atoms is supported by these experimental observations? (1) Gold atoms consist mostly of empty space. In Rutherford's gold foil experiments, some alpha particles were deflected from their original paths but most passed through the foil with no deflection. Which subatomic particles have a mass of approximately 1 atomic mass unit each? (1) proton and electron (2) proton and neutron (3) neutron and positron (4) electron and positron _2.
#The periodic table chemistry test wizar series#